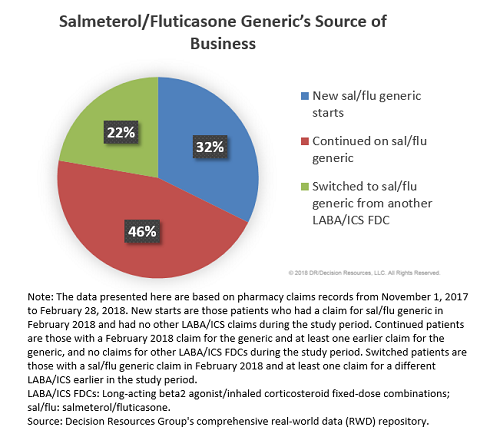
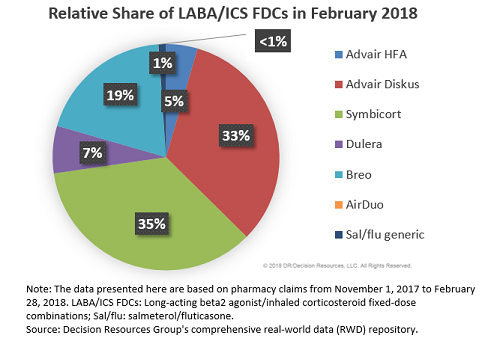
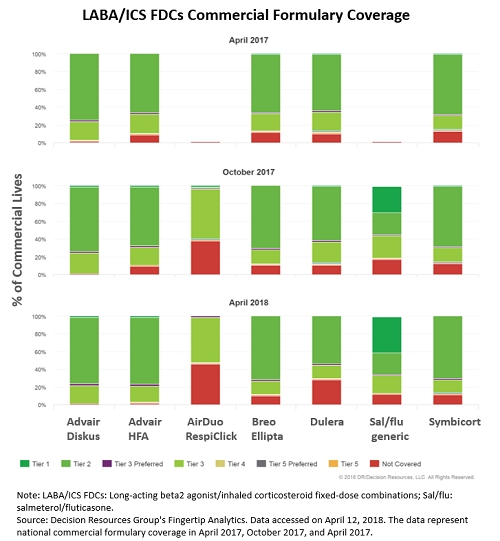
Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire

Teva Pharmaceuticals má v Česku a Slovensku nového generálního ředitele - Chemický a farmaceutický průmysl - www.prumysldnes.cz
EURÓPSKA KOMISIA V Bruseli 28. 1. 2019 COM(2019) 17 final SPRÁVA KOMISIE RADE A EURÓPSKEMU PARLAMENTU PRESADZOVANIE PRÁVA H